
SEAGULL HEALTH
Your health,We care!
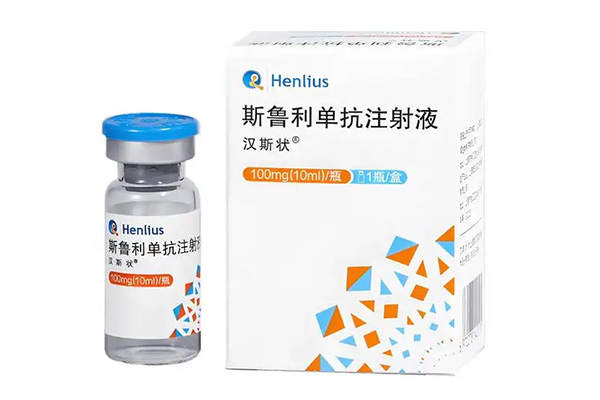
Serplulimab Injection demonstrates definite efficacy and controllable safety whether used in combination with chemotherapy or as monotherapy, with flexible administration regimens and a broad applicable population.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Serplulimab Injection has been approved for marketing in China, the European Union, Indonesia, and nearly 40 other countries and regions worldwide, covering five major indications including microsatellite instability-high (MSI-H) solid tumors, squamous non-small cell lung cancer (sqNSCLC), extensive-stage small cell lung cancer (ES-SCLC), esophageal squamous cell carcinoma (ESCC), and non-squamous non-small cell lung cancer (nsNSCLC).
Indicated for adult patients with unresectable or metastatic microsatellite instability-high (MSI-H) advanced solid tumors, including:
1.Patients with advanced colorectal cancer whose disease progressed after prior treatment with fluoropyrimidine-based drugs, oxaliplatin, and irinotecan;
2.Patients with advanced gastric cancer whose disease progressed after at least two lines of prior therapy and for whom no satisfactory alternative treatment options are available;
3.Patients with other advanced solid tumors whose disease progressed after at least one line of prior therapy and for whom no satisfactory alternative treatment options are available.
4.This indication was granted conditional approval for marketing based on surrogate endpoints. Clinical endpoint data have not yet been obtained, and its efficacy and safety are to be further confirmed in post-marketing studies.
In combination with carboplatin and nab-paclitaxel, this product is indicated for the first-line treatment of unresectable locally advanced or metastatic squamous non-small cell lung cancer (NSCLC).
In combination with carboplatin and etoposide, this product is indicated for the first-line treatment of extensive-stage small cell lung cancer (ES-SCLC).
In combination with fluoropyrimidine-based and platinum-based drugs, this product is indicated for the first-line treatment of PD-L1-positive unresectable locally advanced/recurrent or metastatic esophageal squamous cell carcinoma (ESCC).
This product must be administered under the guidance of a physician experienced in oncology treatment.
Prior to treatment of MSI-H solid tumor patients with this product, the MSI-H status must first be confirmed. A fully validated detection method should be used to confirm the presence of MSI-H before treatment can be initiated.
For the treatment of unresectable locally advanced/recurrent or metastatic esophageal squamous cell carcinoma (ESCC), patients should be selected based on PD-L1 positivity. PD-L1 expression should be assessed using an immunohistochemical (IHC) assay approved by the National Medical Products Administration (NMPA). PD-L1 positivity is defined as a Combined Positive Score (CPS) ≥ 1.
For MSI-H Solid Tumors
The recommended dosage is 3mg/kg, administered as an intravenous infusion once every 2weeks, until disease progression or unacceptable toxicity occurs.
For Squamous Non-Small Cell Lung Cancer (sqNSCLC) or Small Cell Lung Cancer (SCLC)
The recommended dosage is 4.5mg/kg, administered as an intravenous infusion once every 3weeks, until disease progression or unacceptable toxicity occurs.
For Esophageal Squamous Cell Carcinoma (ESCC)
The recommended dosage is 3mg/kg, administered as an intravenous infusion once every 2weeks, until disease progression or unacceptable toxicity occurs.
Patients with hypersensitivity to the active ingredient of Serplulimab Injection or any of the listed excipients are prohibited from using this product.
Adverse reactions with an incidence ≥10% include: anemia, increased aspartate aminotransferase, increased alanine aminotransferase, proteinuria, hypothyroidism, hypoalbuminemia.
The incidence of grade 3 or higher adverse reactions is 30.0%. Those with an incidence ≥1% include: decreased neutrophil count, anemia, decreased white blood cell count, pneumonitis, liver injury, increased gamma-glutamyl transferase, increased aspartate aminotransferase, increased alanine aminotransferase, hyponatremia, hypokalemia, intestinal obstruction, increased blood bilirubin, hyperglycemia or diabetes mellitus, sepsis, abdominal pain, increased conjugated bilirubin, abdominal distension, increased blood creatine phosphokinase.
Adverse reactions with an incidence ≥10% include: anemia, decreased neutrophil count, decreased white blood cell count, decreased platelet count, nausea, fatigue, anorexia, increased alanine aminotransferase, hypothyroidism.
The incidence of grade 3 or higher adverse reactions is 34.9%. Those with an incidence ≥1% include: decreased neutrophil count, decreased white blood cell count, anemia, decreased platelet count, pneumonitis, hyponatremia, decreased lymphocyte count, hyperglycemia or diabetes mellitus, increased alanine aminotransferase.
Pregnancy
There are no available data on the use of this product in pregnant women. Animal studies have shown that PD-1-blocking antibodies have embryo-fetal toxicity. It is known that IgG can cross the placental barrier. As an IgG4 monoclonal antibody, this product may pass from the mother to the developing fetus. Unless the clinical benefit outweighs the potential risk, the use of this product during pregnancy is not recommended.
Lactation
It is currently unknown whether this product is excreted in human milk, or whether it has any effects on breastfed infants or milk production. Since human IgG is secreted into breast milk, this product may pose potential risks to breastfed infants. Therefore, lactating women are advised to discontinue breastfeeding during the treatment period and for at least 6 months after the last dose.
Contraception
Women of childbearing potential should use effective contraceptive measures during the treatment period and for at least 6 months after the last dose.
Fertility
No studies have been conducted to evaluate the effects of this product on fertility in either males or females. Therefore, the impact of this product on male and female fertility remains unknown.
The safety and efficacy of this product in children and adolescents under 18 years of age have not been established.
In the current clinical trials of this product, a total of 535 elderly patients aged ≥ 65 years were enrolled, accounting for 39.8% of all treated patients.
The incidence of all-grade adverse drug reactions was 76.8% in elderly patients versus 73.8% in non-elderly patients;
the incidence of grade 3 or higher adverse drug reactions was 37.8% versus 31.7%;
the incidence of adverse reactions leading to treatment interruption was 30.8% versus 25.7%;
and the incidence of adverse reactions leading to permanent treatment discontinuation was 6.5% versus 5.1%.
No special dose adjustment was performed for elderly patients in clinical studies. Due to the limited number of elderly patients included in the current clinical trials, it is recommended that elderly patients use this product with caution under the guidance of a physician. If use is necessary, no dose adjustment is required.
For more detailed drug information, please consult the official package leaflet.
Serplulimab injection is an anti-tumor drug of···...【详情】
The dosage and administration of serplulimab (···...【详情】
Serplulimab (a PD-1 inhibitor) mainly causes i···...【详情】
Serplulimab (a PD-1 inhibitor) mainly causes i···...【详情】
If any issues arise, please contact us immediately.
Email:haiousales@gmail.com

Serplulimab Injection is contraindicated in patients with known hypersensitivity to serplulimab or any of the excipients in the formulation.
Caution is advised in patients with active autoimmune diseases and organ transplant recipients.
Serplulimab Injection is an anti-PD-1 immune checkpoint inhibitor used for the treatment of specific types of solid tumors.
Store at 2°C to 8°C in a dark place. Do not freeze. The prepared solution should be used within 6 hours.
Copyright 2024 @ haiouhealth.com All right reserved SEAGULL HEALTH



