
SEAGULL HEALTH
Your health,We care!
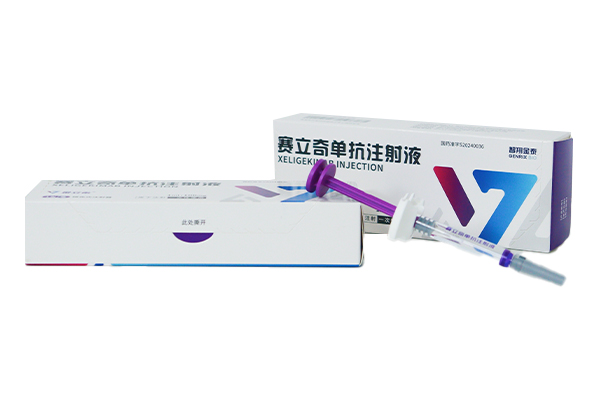
As China's first domestically developed fully human anti-IL-17A targeted biological product, Serelizumab Injection is characterized by rapid onset of action, long-lasting efficacy and favorable safety profile.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy The launch of Xeligekimab Injection has greatly enhanced the accessibility and cost-effectiveness of domestic innovative biological products, providing a high-quality option for long-term standardized treatment to patients with autoimmune diseases.
This product is indicated for the treatment of adult patients with moderate to severe plaque psoriasis who are suitable for systemic therapy or phototherapy.
This product should be used under the guidance and supervision of physicians experienced in the diagnosis and treatment of the indications for this product.
Psoriasis
The recommended dose of this product is 200 mg per administration, to be given at Weeks 0, 2, 4, 6, 8, 10 and 12 respectively, followed by administration once every 4 weeks thereafter.
The 200 mg dose should be administered as two separate injections of 100mg each.
This product should be administered by subcutaneous injection. If possible, injection should be avoided at the psoriatic lesion sites.
Upon approval by healthcare professionals, patients may self-administer this product after receiving training on subcutaneous injection techniques. Physicians shall ensure appropriate follow-up of patients and provide guidance on product use. A comprehensive administration guide is included in the package insert.
Patients with severe hypersensitivity to serelizumab or any of its excipients.
Active infections of clinically significant importance (e.g., active tuberculosis).
The most commonly reported adverse drug reactions (ADR) are upper respiratory tract infections and injection site reactions.
Pregnancy
There are currently no clinical data on the use of this product in pregnant women. Animal studies have not found any direct or indirect adverse effects of the murine analog of serelizumab on pregnancy or fetal development.
However, animal reproductive studies do not fully predict human responses, so it is advisable to avoid the use of this product during pregnancy.
Lactation
It is currently unknown whether serelizumab is secreted in human milk or absorbed systemically after ingestion.
Animal experiments showed that low levels of the murine analog of serelizumab were detected in the milk of ICR mice. In addition, based on the fact that immunoglobulins can be secreted in breast milk,
lactating women should use this product with caution.
Fertility
The effect of serelizumab on human fertility is currently unknown. Animal studies have not found any direct or indirect adverse effects of the murine analog of serelizumab on fertility.
The safety and efficacy of this product in patients under 18 years of age have not been established.
No dosage adjustment is required.
For more detailed drug information, please consult the official package leaflet.
If any issues arise, please contact us immediately.
Email:haiousales@gmail.com

Copyright 2024 @ haiouhealth.com All right reserved SEAGULL HEALTH



