
SEAGULL HEALTH
Your health,We care!
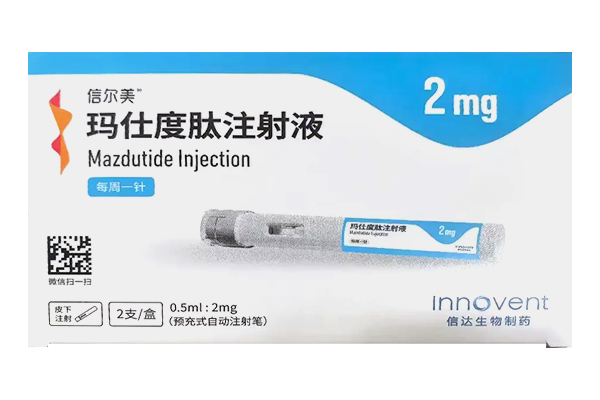
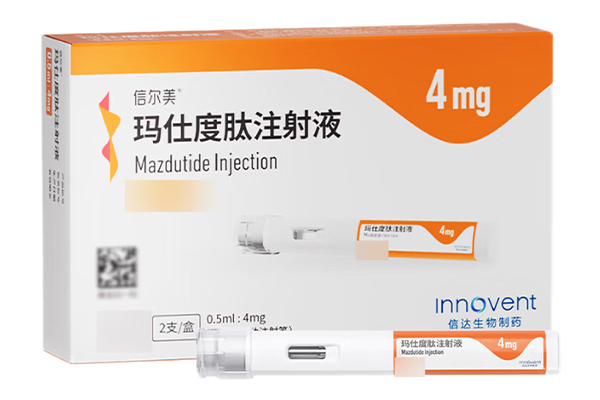
Mazdutide Injection delivers the triple benefits of hypoglycemic effect, weight reduction and liver protection, with a more comprehensive mechanism of action than single-target GLP-1 drugs.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Mazdutide Injection is the world’s only approved glucagon-like peptide-1 receptor (GLP-1R)/glucagon receptor (GCGR) dual agonist. It was approved for marketing by the National Medical Products Administration (NMPA) in June 2025, with its indication for diabetes approved in October of the same year.
This product is indicated for long-term weight management in adult patients, as an adjunct to a reduced-calorie diet and increased physical activity, in those with an initial body mass index (BMI) of:
≥28kg/m² (obesity), or ≥24kg/m² (overweight) and accompanied by at least one weight-related comorbidity (e.g., hyperglycemia, hypertension, dyslipidemia, fatty liver disease, obstructive sleep apnea syndrome, etc.).
The recommended initial dose of mazdutide is 2mg, administered subcutaneously once weekly.
After 4 weeks of recommended administration, the dose should be increased to 4mg, administered subcutaneously once weekly. If further weight improvement is required, the dose may be increased to 6mg once weekly after receiving treatment with the current dose for at least 4 weeks.
The recommended maintenance dose of mazdutide is 4mg or 6mg, administered subcutaneously once weekly. If the 6mg dose is not tolerated, the dose may be considered for reduction back to 4mg for maintenance.
Hypersensitivity to the active ingredient of this product or any of its excipients.
Personal history or family history of medullary thyroid carcinoma (MTC), or diagnosis of multiple endocrine neoplasia type 2 (MEN2).
In overweight or obese patients receiving mazdutide for long-term weight management, the adverse reactions with an incidence rate ≥ 2% and higher than that in the placebo group are as follows:
Mazdutide 4 mg group: The most common adverse reactions are nausea, diarrhea, and vomiting; other common adverse reactions include abdominal distension, abdominal pain, injection site reactions, dizziness, gastroesophageal reflux disease, fatigue, belching, constipation, hiccups, and dyspepsia.
Mazdutide 6 mg group: The incidence of adverse reactions is generally higher than that in the 4 mg group, with the main adverse reactions being nausea, diarrhea, and vomiting; other adverse reactions include abdominal distension, abdominal pain, injection site reactions, dizziness, gastroesophageal reflux disease, fatigue, belching, constipation, hiccups, and dyspepsia.
Pregnancy
The effects of mazdutide on pregnant women are not yet clear; therefore, this product is contraindicated in pregnant women. If a patient is planning to become pregnant or is already pregnant, mazdutide treatment should be discontinued. Given the long half-life of this product, mazdutide should be discontinued at least 3 months prior to planned pregnancy.
Lactation
It is not known whether mazdutide is excreted in human milk. Risks to neonates/infants cannot be excluded. This product must not be used during lactation.
Women of Childbearing Potential
Women of childbearing potential receiving mazdutide treatment are recommended to take contraceptive measures.
Fertility
The effects of mazdutide on human fertility are not yet clear.
The safety and effectiveness of mazdutide in pediatric and adolescent patients under 18 years of age have not been established.
In clinical trials of mazdutide for the treatment of obese or overweight patients, the number of patients aged 65 years and above was limited (N=3), and thus the safety and effectiveness of mazdutide in geriatric patients cannot be determined.
For more detailed drug information, please consult the official package leaflet.
If any issues arise, please contact us immediately.
Email:haiousales@gmail.com

Copyright 2024 @ haiouhealth.com All right reserved SEAGULL HEALTH



