
SEAGULL HEALTH
Your health,We care!
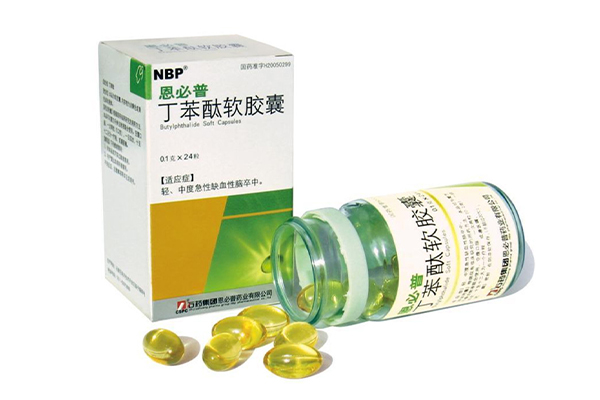
The launch of Butylphthalide Soft Capsules has not only been widely applied in China, but also passed on-site inspections by the U.S. FDA, initiated Phase Ⅲ clinical trials overseas and obtained Orphan Drug Designation.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Butylphthalide can alleviate cerebral cell damage caused by acute ischemic stroke and further improve the neurological deficit symptoms of patients through multiple mechanisms, including improving microcirculation in the cerebral ischemic area, promoting the establishment of collateral circulation, and protecting mitochondrial function.
It is indicated for the treatment of mild to moderate acute ischemic stroke.
Based on the medication method from existing clinical studies, this product can be used in combination with Compound Salvia Miltiorrhiza Injection. It should be administered orally on an empty stomach.
The recommended dosage is 2 capsules (0.2g) each time, three times daily. A treatment course lasts for 20 days, or follow the doctor’s advice.
This product is contraindicated in the following patients:
Patients hypersensitive to this product.
Patients with severe bleeding tendency.
Adverse reactions to this product are rare. The main reaction is mild and transient elevation of aminotransferase, which can return to normal after drug withdrawal according to follow-up observation cases. Nausea, abdominal discomfort and psychiatric symptoms are occasionally reported.
The efficacy and safety of this product in pregnant and lactating women have not been studied.
The efficacy and safety of this product in children have not been established.
See Dosage and Administration.
For more detailed drug information, please consult the official package leaflet.
If any issues arise, please contact us immediately.
Email:haiousales@gmail.com

Copyright 2024 @ haiouhealth.com All right reserved SEAGULL HEALTH



